Aims: Cranioplasty and facial bone reconstruction are surgical procedures used for craniofacial defects following trauma or neurosurgical procedures like decompressive craniectomy; with the aid of prosthesis made from several materials. Implant customized manufacturing for craniofacial defects allows for a precise and anatomical reconstruction in a shorter operating time but the prosthesis production is often costly and requires complex intraoperative processes. In this case report we introduce a simple, low-cost method for prosthesis manufacturing.
Case Presentation: We present the case of a 30-year-old male patient with skull and facial bone comminuted fracture who received three-dimensional (3D) printing customized polymethyl methacrylate (PMMA) prosthesis for facial bone and frontal bone defects reconstruction. Post-operative 10 months follow-up showed excellent patient, family, and surgeon satisfaction at a fraction of the cost associated with commercially available implants.
Conclusion: The use of customized 3D PMMA prosthesis could be an attractive option to all patients undergoing cranioplasty.
Cranioplasty; Craniofacial Defects; Polymethylmethacrylate Resin; PMMA Prosthesis; 3D Printing
Craniofacial defects usually occur after trauma, neurosurgical procedures like decompressive craniectomy, tumor resections, infection or congenital defects. The purpose of cranial vault repair is to protect the underlying brain tissue, to reduce any localized pain and patient anxiety, and improve cranial aesthetics [1]. Cranioplasty and facial bone reconstruction are frequent neurosurgical and plastic surgical procedure achieved with the aid of prosthesis made from materials such as: titanium, autologous bone, ceramics and polymers [2]. Prosthesis production is often costly and requires complex intraoperative processes. Implant customized manufacturing for craniofacial defects allows for a precise and anatomical reconstruction in a shorter operating time compared to other conventional techniques. We introduce a simple, low-cost method for prosthesis manufacturing [3, 4].
One 30-year-old male patient who was generally healthy before suffered from traffic accident with loss of consciousness and face on the ground found by passenger at night. Physical examination revealed open fracture over left frontal region with brain tissue exposure. He was sent to our hospital for rescue. Image studies revealed findings as below:
- Head injury with comminuted fracture of skull and facial bone complicated with acute epidural and subarachnoid hemorrhage over frontal region, left and brain tissue loss.
- Facial bone fracture of infraorbital rim, maxillary buttress right
- Herniated intervertebral disc of C5-C6 with myeloradiculopathy
- Open fracture of patella, right
- Fracture of humerus, right
He received life-saving surgery of left frontal skull, nasal bone, bilateral orbital bone fractured bone removal with facial wound debridement and primary closure and left frontotemporal craniectomy with removal of extradural hematoma immediately (Figure 1).
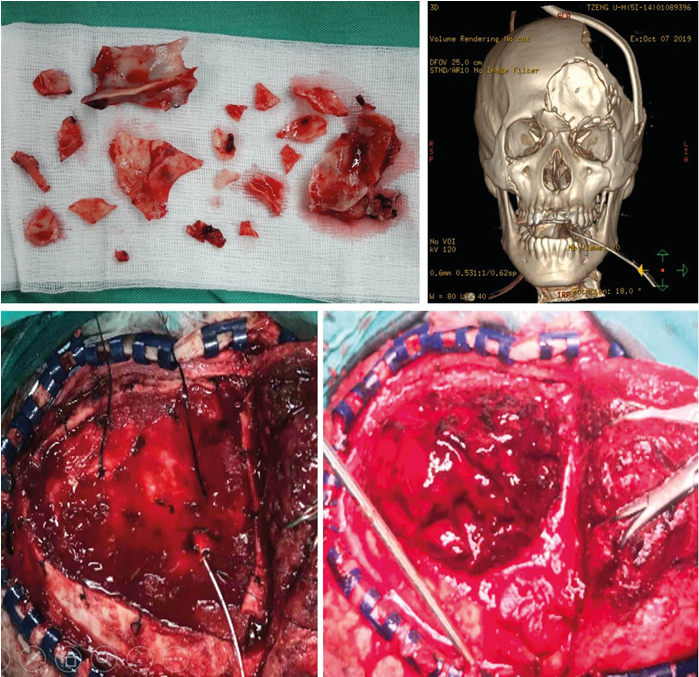
Figure 1:
He also received anterior approach with C5-C6 cervical discectomy and interbody fusion with cervical artificial disc for C5-C6 spinal cord compression and bilateral neuroforaminal impingement a few days later. open reduction and internal fixation for right patella and humerus fracture were also performed.
Cranioplasty for left frontotemporal craniectomy defect was performed with Autogenous bone flap. For facial bone fracture, firstly we performed open reduction and internal fixation via Gillies approach with microplates and screws. The three-dimensional (3D) printing technique for facial bone and frontal bone defects reconstruction was also arranged. A digital prosthesis model is designed and manufactured with the aid of a 3D computed tomography. Both the data of large sized cranial defects and the prosthesis are transferred to a 3D printer to obtain a physical model in poly-lactic acid which is then used in a laboratory to cast the final customized prosthesis in polymethyl methacrylate (PMMA) (Figure 2).
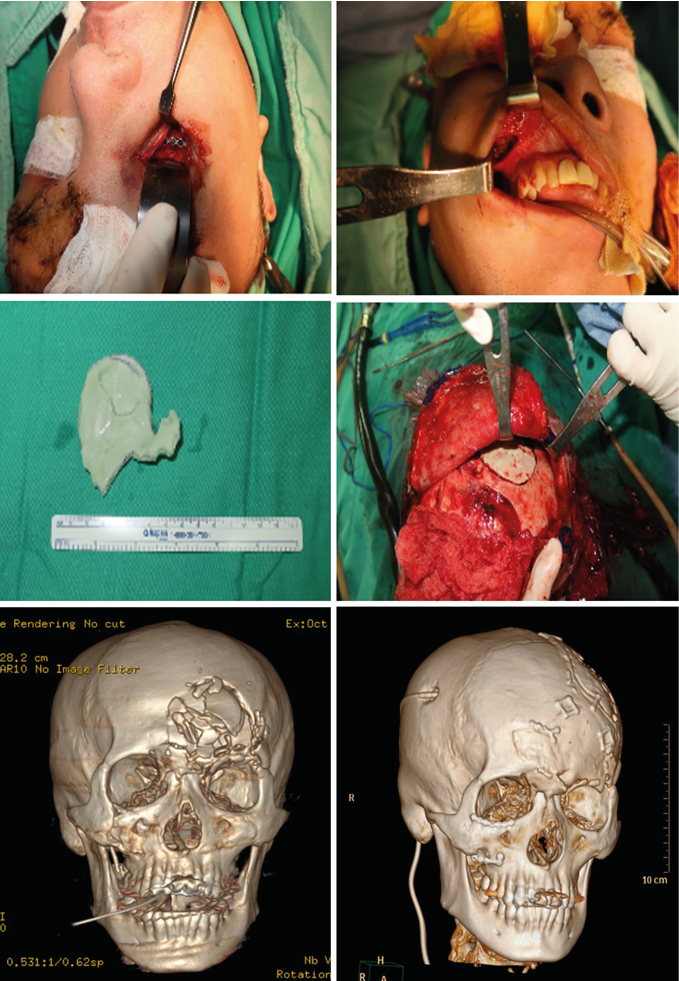
Figure 2.
The patient recovered gradually and smoothly after reconstructive surgery. Rehabilitation program was arranged and finally he discharged from our hospital 2 months later after the date of traffic accident. Follow-up recovery condition about half-a-year later showed very excellent cosmetic result compared with appearance before the accident (Figure.3).

Figure 3.
Cranioplasty implants should be widely available, low in cost, and customized or easy to mold during surgery. Although autologous bone remains the first choice for repair, it cannot always be used due to infection, fragmentation, bone resorption, or other causes, which led to use of synthetic alternatives. Since this case treated at our Hospital presented with fragmented cranial fractures, bone preservation was unachievable.
Hand-manufacturing of a prosthetic implant can only be carried out if the original cranial bone fragment is available [1]. The most frequently used allogenic material for cranial reconstructions with long-term results is polymethylmethacrylate (PMMA). Three-dimensional printing technology has allowed the production of increasingly popular customized, prefabricated implants. PMMA prosthesis manufacturing by hand has been used since 1970 using various processes [5-8], but these methods have been overtaken by computer assisted design and manufacturing techniques (CAD/CAM) which consist of using images of the cranial defect and manufacturing the PMMA prostheses with a 3D printer [9–15]. The CAD/CAM technique described by Caro-Osorio & Cols. [16] in 2013 is no longer an expensive method. The ideal implant material should have the following characteristics: it should be able to adjust to the cranial defect, it must achieve a full defect coverage, full biocompatibility, inertness, non-thermal conductor, radiolucent, non-magnetic, light, rigid, easily placed and low-cost [9, 17].
The CT scan data is stored in the standard format DICOM (Digital Images and Communications in Medicine) which allows generating an interface between the medical equipment and any other device to visualize the images. Through the DICOM viewer which generated a three-dimensional reconstruction of all the CT cross-sectional images. A bone filter is applied in order to only observe the bone structure, achieved by taking as reference its attenuation degree. The implant is generated using the software of the computer-aided design. Finally, the data is exported in a stereolithography extension file (STL) and fed to the printer [1].
A 3D printer is used to print out a PLA prosthesis from the STL file using a fused deposition model. Once the printing process is finished, the scaffolds are removed with a low-speed motor and a carbide bur and the adjustment is verified. The prosthesis is placed in metal containers to obtain two plaster impressions (an internal and an external surface impression). A transparent PMMA OPTI-CRYLR is poured into the space created by the internal plaster impression and pressed with the external mold. The casts are opened and the prosthesis is revealed after a 25-min polymerization time. The prosthesis is then cut, perforated, and polished with a low-speed motor [1]. (Figure.4)

Figure 4.
The use of customized 3D PMMA prosthesis was associated with excellent patient, family, and surgeon satisfaction at follow-up at a fraction of the cost associated with commercially available implants. This technique could be an attractive option to all patients undergoing cranioplasty.
- De La Peña (2018) 3D Printing in Medicine 4: 4.
- Neumann A, Kevenhoerster K (2011) Biomaterials for craniofacial reconstruction. GMS Curr Top Otorhinolaryngol Head Neck Surg 8. [Crossref]
- Marchac D, Greensmith A (2008) Long-term experience with methylmethacrylate cranioplasty in craniofacial surgery. J Plast Reconstr Aesthet Surg 61: 744-753. [Crossref]
- Moreira-Gonzalez A, Jackson IT, Miyawaki T, et al. (2003) Clinical outcome in cranioplasty: critical review in long-term follow-up. J Craniofac Surg 14:144-153. [Crossref]
- Dumbrigue HB, Arcuri MR, LaVelle WE, et al. (1998) Fabrication procedure for cranial prostheses. J Prosthet Dent 79:229-131. [Crossref]
- Jordan RD, White JT, Schupper N (1978) Technique for cranioplasty prosthesis fabrication. J Prosthet Dent 40: 230-233. [Crossref]
- Origitano TC, Izquierdo R, Scannicchio LB (1995) Reconstructing complex cranial defects with a preformed cranial prosthesis. Skull Base Surg 5: 109-116. [Crossref]
- Yamamoto Y, Mendel E, Raffel C (1997) Acrylic cranioplasty with alginate molding: technical note. Neurosurgery 41: 305-309. [Crossref]
- Chiarini L, Figurelli S, Pollastri G, et al. (2004) Cranioplasty using acrylic material: a new technical procedure. J Craniomaxillofac Surg 32: 5-9. [Crossref]
- Chrzan R, Urbanik A, Karbowski K, et al. (2012) Cranioplasty prosthesis manufacturing based on reverse engineering technology. Med Sci Monit 18: MT1-6. [Crossref]
- Dean D, Min KJ, Bond A (2003) Computer aided design of large-format prefabricated cranial plates. J Craniofac Surg 14: 819-832. [Crossref]
- Gerber N, Stieglitz L, Peterhans M, (2010) Using rapid prototyping molds to create patient specific polymethylmethacrylate implants in cranioplasty. Conf Proc IEEE Eng Med Biol Soc 3357-3360. [Crossref]
- Goh RC, Chang CN, Lin CL, Lo LJ. Customised fabricated implants after previous failed cranioplasty. J Plast Reconstr Aesthet Surg 63: 1479-1484. [Crossref]
- Lee SC, Wu CT, Lee ST, et al. (2009) Cranioplasty using polymethyl methacrylate prostheses. J Clin Neurosci 16: 56-63. [Crossref]
- Rotaru H, Stan H, Florian IS, et al. (2012) Cranioplasty with custom-made implants: analyzing the cases of 10 patients. J Oral Maxillofac Surg 70: 169-176. [Crossref]
- Caro-Osorio E, De la Garza-Ramos R, Martínez-Sánchez SR, et al. (2013) Cranioplasty with polymethylmethacrylate prostheses fabricated by hand using original bone flaps: technical note and surgical outcomes. Surg Neurol Int 4: 136. [Crossref]
- Blake DP (1994) The use of synthetics in cranioplasty: a clinical review. Mil Med 159: 466-469. [Crossref]




