Background: Proliferative Diabetic Retinopathy (PDR) can be treated with intra-vitreal injection of anti-VEGF compounds. It can be expected that the study of the effects of this treatment on the diameter and oxygen saturation of larger retinal vessels may contribute to our knowledge about the pathophysiology of PDR.
Methods: Seventeen patients referred with PDR and neovascularizations at the optic disk were subjected to retinal oximetry before and (mean ± SD) 6.9 ± 3.8 days after intra-vitreal injection with anti-VEGF compound. Changes in diameter and oxygen saturation in the larger retinal vessels before and after treatment were assessed.
Results: At the follow-up examinations the diameter of retinal arterioles had decreased significantly from (mean ± SD) 118.7 ± 13.9 µm to 109.5 ± 18.7 µm (p<0.01), whereas there was no significant change in the diameter of retinal venules (p=0.24). There was no significant changes in the oxygen saturation in retinal arterioles (from 97.3 ± 6.1 % to 98.0 ± 6.3 %, p=0.44) or in retinal venules (from 63.2 ± 12.1 % to 62.3 ± 10.4 %, p=0.66).
Conclusions: Regression of retinal neovascularizations after treatment of proliferative diabetic retinopathy is related to constriction of retinal arterioles but not to changes in oxygen saturation in the larger retinal vessels.
Diabetic retinopathy; retina – Medical therapies; Techniques of retinal examination; Retinal degeneration associated with systemic disease; Retinal pathology / research
Diabetic retinopathy is accompanied with changes in retinal blood flow that present as vasodilatation and hyper permeability in the macular area and occlusion of the capillary bed in the retinal periphery [1,2]. The ischemic and hypoxic peripheral retina releases Vascular Endothelial Growth Factor (VEGF) that stimulates the development of Proliferative Diabetic Retinopathy (PDR) [3]. The new vessels in PDR can be treated by Pan Retinal Photocoagulation (PRP) [4], and the combined increase in the oxygen supply and reduction in the metabolic demand induced by this treatment may explain the observed subsequent increase in the oxygen saturation but constriction of retinal arterioles [5,6].
Intra-vitreal injection with anti-VEGF drugs is a new tool for inhibiting neovascularizations in PDR, either administered alone or adjunctive to PRP [7,8]. It can be expected that the study of how changes in the oxygen saturation and diameter of the larger retinal vessels after anti-VEGF treatment differ from the changes in these parameters after PDR can contribute to understanding the pathophysiology and the therapeutic effects on PDR. Anti-VEGF treatment has been shown not to affect oxygen saturation in larger retinal vessels after repeated injections for 12 and 52 weeks in PDR, but this finding may reflect accumulated effects of the treatment or adaptive mechanisms occurring independently of the regression of the neovascularizations [9].
Therefore, the oxygen saturation and diameter of larger retinal vessels were studied before and shortly after one intra-vitreal injection with anti-VEGF compound that induced regression of neovascularizations in PDR.
Design
Prospective observational study of oxygen saturation and diameter of larger retinal vessels before and (mean ± SD) 6.9 ± 3.8 days after intravitreal injection with an anti-VEGF compound (aflibercept or ranibizumab) in seventeen patients with Proliferative Diabetic Retinopathy (PDR) and new vessels at the optic disk.
Study population
Twenty-eight successive patients diagnosed with PDR and neovascularizations at the optic disk covering an area of more than one disk diameter with clear optical media and distinguishable peripapillar vascular arcades to all four retinal quadrants examined at the Department of Ophthalmology, Aarhus University Hospital between January 2015 and December 2017 were included.
The patients were interviewed about previous ocular and systemic disease and underwent a routine examination consisting of measurement of best corrected visual acuity followed by induction of mydriasis with tropic amide (1%) and phenylephrine (10%) eye drops, fundus photography (Canon CF 60Z fundus camera, Canon, Amstelveen, the Netherlands), slit lamp examination and retinal oximetry (Oxymap model T1, version 2.4.2, V.7249 V2, Oxymap, Reykjavik, Iceland). Fundus photography and oximetry were planned within one week in order to monitor the regression of the new vessels before initiating panretinal photocoagulation. Clinical and demographic characteristics of the included patients are shown in Table 1.
Table 1. Clinical characteristics of the study population. T1D = type 1 diabetes. T2D = type 2 diabetes.
|
Right/left (n)
|
13/4
|
|
Males/females (n)
|
13/4
|
|
Age (years ± SD)
|
50.0 ± 14.0
|
|
T1D/T2D (n)
|
6/11
|
|
Diabetes duration (years ± SD)
|
16.2 ± 12.1
|
Oximetry
Retinal oximetry was performed using an Oxymap oximeter (Oxymap model T1, version 2.4.2, V.7249 V2, Oxymap, Reykjavik, Iceland). Images were obtained at five photographic fields, i.e one centered at the fovea, two centered at the optic disk and two with respectively the upper and lower temporal arcade in the image center.
The fundus photograph centered on the optic disk which was considered to have the best image quality was selected for the analysis. In order to standardize the measurement of vessel saturation and diameter, an area between two circles concentric with the optic disk was defined (Figure 1). An inner circle with a diameter 30 pixels larger than the disk radius was defined manually using the inbuilt marking tool in the oximeter software, and an outer circle was defined with a diameter one disk larger than that of the optic disk. Within this circular zone, oxygen saturation values were obtained on the longest unbranched segment of the four larger arterioles and venules supplying respectively the lower temporal, lower nasal, upper nasal and upper temporal quadrant, however excluding vessel segments shorter than 50 pixels and with a diameter smaller than 60 micrometers. The saturation and diameter of the four studied arterioles and venules were averaged to one value for each of the arterial and venous saturations and diameters for each patient. Image selection and subsequent analysis were all performed by the same person (INS).
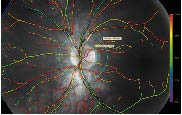
Figure 1. Example of a fundus photograph obtained with the oximeter. The marking of the retinal vessels shows high saturation (red) in the arterioles and low saturation (green) in the venules. The border of the optic nerve head could be delimited by a circle with a diameter of 226 pixels from which two circles with respectively the double and a 30 pixel larger diameter were defined. The longest unbranched arteriolar and venular segments to the four quadrants between the two concentrical circles are delimited with white lines
Treatment
In order to reduce the risk of vitreous haemorrhage the patients received one injection with intravitreal ranibizumab 0.5mg (n=4) or with intravitreal aflibercept 2mg (n=24), which was planned to precede pan retinal photocoagulation. The choice of anti-VEGF compound depended on national guidelines at the time of treatment.
Follow-up
Follow-up oximetry examinations were planned to be within one week following injection with anti-VEGF medication. At these examinations oximetry was not possible or insufficient in 8 patients who received treatment with aflibercept and 1 patient receiving treatment with ranibizumab. Furthermore, due to individual preferences, follow-up was performed later than three weeks after the injection in two patients, which left seventeen patients to be included. Among these fourteen had received treatment with aflibercept and three with ranibizumab. In the included patients, the follow-up time was (mean ± SD) 6.9 ± 3.8 (range 2–17) of the injection.
Data analysis/Statistics
Based on previous experience it could be calculated that with a power of 0.8 and a p-value at 0.05, the inclusion of 17 patients was sufficient to detect a 13 % change in the diameter and oxygen saturation of larger retinal vessels [10].
Microsoft Excel 2007 was used for data storage and all statistical analyses and graphical presentation of the results were conducted using GraphPad Prism (version 5.00, GraphPad Software, San Diego California USA). Data was tested for normality and presented as mean ± Standard Deviation (SD). Changes from before to after treatment were tested using Student’s paired t-test.
Figure 2 shows a representative example of the observed regression of neovascularizations seven days after one anti-VEGF injection and Figure 3 the measured diameters and oxygen saturations before and after the anti-VEGF injection in all patients.
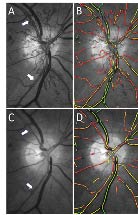
Figure 2. An example of the observed clinical effect. A: Neovascularizations at the optic disk. The arrows point to the segment of the temporal arterioles where diameter and oxygen saturation were measured. B: Same image with colour coding of oxygen saturation. The low oxygen saturation in venules is marked as green whereas the high oxygen saturation in the arterioles (and the parts of the neovascularization that can be resolved) is marked as red. C: Regression of the new vessel seven days after treatment with 2 mg aflibercept. The arterioles are seen to have been narrowed. D: Same image with unchanged colour coding of the oxygen saturation
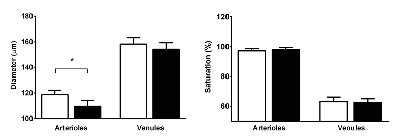
Figure 3. The measured diameters and saturations in retinal arterioles and venules before (white columns) and after (black columns) single injection with anti-VEGF compound. Error bars indicate SEM
At the follow-up examination the diameter of the larger retinal arterioles (mean ± SD) were significantly (p<0.01) reduced by approximately 8% (from 118.7 ± 13.9 to 109.5 ± 18.7 µm), whereas there was no significant change in the diameter of venules (from 158.3 ± 19.2 to 154.2 ± 21.1 µm) and no significant change in the oxygen saturations in neither the arterioles (from 97.3 ± 6.1 % to 98.0 ± 6.3 %, p=0.44) nor in the venules (from 63.2 ± 12.1 % to 62.3 ± 10.4 %, p=0.66).
A recalculation of changes in diameters and oxygen saturations after the exclusion of the patients treated with ranibizumab resulted in the same conclusions. There were no significant differences in vascular diameter or oxygen saturation between patients with type 1 and type 2diabetes, neither before nor after treatment (p > 0.15 for all comparisons).
A number of studies have shown that repeated injections with anti-VEGF compound can reduce the size of neovascularizations in Proliferative Diabetic Retinopathy (PDR) and reduce the risk of vitreous haemorrhage from these lesions [7,9,11]. The effect is assumed to be related to the inhibition of VEGF released by hypoxic retinal tissue, but it is unknown to what extent the vascular abnormalities assumed to underlie the release of VEGF are affected by the treatment. This might be further elucidated by studying changes in the diameter and oxygen saturation of larger retinal vessels in parallel with the regression of the neovascularizations occurring shortly after the first anti-VEGF injection. The diameter and oxygen saturation in the larger retinal vessels can be assessed by retinal oximetry and during the recent years this method has provided important new evidence about the pathophysiology of diabetic retinopathy [12,13]. The oxygen saturation is an independent risk factor for the severity of diabetic retinopathy with the same predictive value for retinopathy grade as metabolic regulation and blood pressure [14]. When retinopathy has progressed to Proliferative Diabetic Retinopathy (PDR) the diameter of the larger retinal vessels is increased and the oxygen saturation in the neovascularizations is arterial which suggest that these vessels act as shunts to bypass areas of capillary occlusion [15,16]. After treatment with PRP the diameter of the dilated larger retinal arterioles decreases, whereas the oxygen saturation increases further [6,17]. Although the observed increase in the oxygen saturation may partially be due to a reduction in the linear velocity of the blood, the findings suggest that the effect of pan retinal photocoagulation is related to the normalization of the arterial diameter rather than to a change in the oxygen saturation in the larger retinal vessels [18].
In the present study the diameter and oxygen saturation in the larger retinal vessels were studied shortly after the first intra-vitreal injection with anti-VEGF compound which ensured the monitoring of effects on vessel diameter and oxygen saturation simultaneously with the regression of the neovascularizations. Furthermore, the effect was not influenced by previous anti-VEGF injections or adaptations in flow and metabolism that may occur over months [19]. Although the study only showed changes in diameter of retinal arterioles, it cannot be excluded that a change in the diameter of retinal venules or the oxygen saturations might have been detected if more patients had been included. However, this does not invalidate the finding of a significant constriction of retinal arterioles shortly after one anti-VEGF injection as the most salient change in diameter and oxygenation of the larger vessels.
The diameter of retinal arterioles is auto regulated which results in vasodilatation when the blood flow is insufficient to support retinal metabolism. The observed dilatation of retinal arterioles in diabetic patients can therefore be interpreted as an auto regulatory response attempting to increase the blood flow to retinal tissue which is hypoxic secondary to capillary occlusion. However, the lack of increase in the flow through the occluded capillaries results in an increase in the hydrostatic pressure in the remaining perfused vessels. This can explain the shunting of blood to bypass the capillary bed which increases the oxygen saturation in the larger retinal venules with increasing retinopathy grade [13,20,21]. The constriction of retinal arterioles after pan retinal photocoagulation can be considered to be a result of a reduction in the metabolic drive to dilate the arterioles because the amount of hypoxic retinal tissue is reduced and due to facilitated diffusion of oxygen from the choroidal vessels [5,6,17]. This in turn reduces the hydrostatic pressure in the arterioles and the need for developing intra-retinal shunts and retinal neovascularizations which is observed as the beneficial clinical effect of the treatment. However, intra-vitreal injections of anti-VEGF compound are not accompanied with destruction of retinal tissue and therefore the constriction of retinal arterioles after anti-VEGF treatment can be assumed to have a different background than the constriction observed after pan retinal photocoagulation. Anti-VEGF treatment has been shown to contract retinal arterioles in different retinal diseases such as neovascular age-related macular degeneration and diabetic maculopathy [22-24]. The effect may both be a result of blocking of signals from the hypoxic retinal areas that stimulate dilatation of the arterioles or a result of reduced demand for blood supply secondary to improved retinal metabolism or capillary flow. Reduced arterio-venous shunting might be expected to also reduce the oxygen saturation in the retinal venules, but this has not been observed, neither after one injection as performed in the present study nor after repeated injections as reported previously [9]. It is possible that a change in the true oxygen saturation may have been masked by opposite changes in blood flow since it has recently been shown that the oxygen saturation in larger retinal vessels correlates inversely with the linear velocity of the blood [18]. Alternatively, the changes in perfusion after treatment may have balanced the changes in metabolic demand to maintain oxygen saturation unchanged.
In conclusion, the study confirms that the beneficial effect of pan retinal photocoagulation and anti-VEGF treatment on retinal neovascularizations in proliferative diabetic retinopathy is related to constriction of retinal arterioles but not to changes in the oxygen saturation of the larger retinal vessels. This adds to our understanding of the pathophysiology of diabetic retinopathy and points to new targets of intervention for improving treatment of the disease.
The skillful assistance of nurse photographers Helle Hedegaard and Tina Bjerre is gratefully acknowledged.
The VELUX Foundation. The Toyota Foundation.
None
- Kristinsson JK, Gottfredsdóttir MS, Stefánsson E (1997) Retinal vessel dilatation and elongation precedes diabetic macular oedema. Br J Ophthalmol. 81: 274-278. [Crossref]
- Bek T (2009) Inner retinal ischaemia: current understanding and needs for further investigations. Acta Ophthalmol. 87: 362-367. [Crossref]
- Bek T (2013) Regional morphology and pathophysiology of retinal vascular disease. Prog Ret Eye Res. 36: 247-259. [Crossref]
- The Diabetic Retinopathy Study Research Group (1981) Photocoagulation treatment of proliferative diabetic retinopathy. Clinical application of Diabetic Retinopathy Study (DRS) findings, DRS Report Number 8. Ophthalmology. 88: 583-600. [Crossref]
- Stefánsson E, Machemer R, de Juan E, McCuen BW, Peterson J (1992) Retinal oxygenation and laser treatment in patients with diabetic retinopathy. Am J Ophthalmol. 113: 36-38. [Crossref]
- Jørgensen C, Bek T (2014) Increasing oxygen saturation in larger retinal vessels after photocoagulation for diabetic retinopathy. Invest Ophthalmol Vis Sci. 55: 5365-5369. [Crossref]
- Sivaprasad S, Prevost AT, Vasconcelos JC, Riddell A, Murphy C, et al. (2017) Clinical efficacy of intravitreal aflibercept versus panretinal photocoagulation for best corrected visual acuity in patients with proliferative diabetic retinopathy at 52 weeks (CLARITY): a multicentre, single-blinded, randomised, controlled, phase 2b, non-inferiority trial. Lancet. 389: 2193-2203. [Crossref]
- Ali W, Abbasi KZ, Raza A (2018) Panretinal Photocoagulation Plus Intravitreal Bevacizumab Versus Panretinal Photocoagulation Alone for Proliferative Diabetic Retinopathy. J Coll Physicians Surg Pak. 28: 923-927. [Crossref]
- Nicholson L, Crosby-Nwaobi R, Vasconcelos JC, Prevost AT, Ramu J, et al. (2018) Mechanistic evaluation of panretinal photocoagulation versus aflibercept in proliferative diabetic retinopathy: CLARITY substudy. Invest Ophthalmol Vis Sci. 59: 4277-4284. [Crossref]
- Dong Y, Lin L, Yan H, Fu Y, Zong Y, et al. (2016) Shifts in retinal vessel diameter and oxygen saturation in Chinese type 2 diabetes mellitus patients. BMC Ophthalmol. 16: 43. [Crossref]
- Arevalo JF, Lasave jf, Wu L, Diaz-Llopis M, Alezzandrini AA, et al. Intravitreal bevacizumab for proliferative diabetic retinopathy. Results from the pan-american collaborative retina study group (PACORES) at 24 months of follow-up. Retina. 2017; 334-343. [Crossref]
- Hardarson SH, Harris A, Karlsson RA, Halldorsson GH, Kagemann L, et al. (2006) Automatic retinal oximetry. Invest Ophthalmol Vis Sci. 47: 5011-5016. [Crossref]
- Jørgensen CM, Hardarson SH, Bek T (2014) The oxygen saturation in retinal vessels from diabetic patients depends on the severity and type of vision-threatening retinopathy. Acta Ophthalmol. 92: 34-39. [Crossref]
- Bek T, Stefánsson E, Hardarson SH (2019) Retinal oxygen saturation is an independent risk factor for the severity of diabetic retinopathy. Br J Ophthalmol. 103: 1167-1172. [Crossref]
- Pedersen L, Jeppesen P, Knudsen ST, Poulsen PL, Bek T (2014) Improvement of mild retinopathy in type 2 diabetic patients correlates with narrowing of retinal arterioles. A prospective observational study. Graefes Arch Clin Exp Ophthalmol. 252: 1561-1567. [Crossref]
- Bek T (2018) Arterial oxygen saturation in neovascularizations in proliferative diabetic retinopathy. Retina. 38: 2301-2308. [Crossref]
- Gottfredsdóttir MS, Stefánsson E, Jónasson F, Gíslason I (1993) Retinal vasoconstriction after laser treatment for diabetic macular edema. Am J Ophthalmol. 115: 64-67. [Crossref]
- Jeppesen SK, Bek T (2019) The Retinal Oxygen Saturation Measured by Dual Wavelength Oximetry in Larger Retinal Vessels is Influenced by the Linear Velocity of the Blood. Curr Eye Res. 44: 46-52. [Crossref]
- Bek T, Jørgensen CM (2016) The Systemic Blood Pressure and Oxygen Saturation in Retinal Arterioles Predict the Effect of Intravitreal Anti-VEGF Treatment on Diabetic Maculopathy. Invest Ophthalmol Vis Sci. 57: 5429-5434. [Crossref]
- Hammer M, Vilser W, Riemer T, Mandecka A, Schweitzer D, et al (2009) Diabetic patients with retinopathy show increased retinal venous oxygen saturation. Graefes Arch Clin Exp Ophthalmol 247: 1025-1030. [Crossref]
- Hardarson SH, Stefánsson E (2012) Retinal oxygen saturation is altered in diabetic retinopathy. Br J Ophthalmol. 96: 560-563. [Crossref]
- Tetikoglu M, Kurt MM, Sagdik HM et al. Retrospective analysis of the effect of aflibercept loading dose on the retinal vessel diameters in patients with treatment-naive neovascular AMD. Cutan Ocul Toxicol 2018; 37/1: 84-89. [Crossref]
- Kurt MM, Cekic O, Akpolat C et al. Effects of intravitreal ranibizumab and bevacizumab in the retinal vessel size in diabetic macular edema. Retina 2018; 38/6: 1120-1126. [Crossref]
- Consigli A, Papanastasiou A, Roquelaure D et al. Changes in Retinal Vascular Caliber after Intravitreal Aflibercept Treatment for Diabetic Macular Oedema. Klin Monbl Augenheilkd. Epub ahead of print 2 August 2018. DOI: 10.1055/a-0630-1664. [Crossref]



